Calibration & Verification - GMPUSP GC41,1058, 1251
Product Details:
Product Description
Calibration & Verification - GMPUSP GC41,1058, 1251
Verification of balance as per USP revision requirement. It is Electronic Generated Document.
USP requirements are:
- Calibration: GMP requirement
- Repeatability: Random Error
- Accuracy: Systematic Error
- Risk Assessment: Reproducibility & Safety
Revision USP and GWP altogether fulfills the concern requirements:
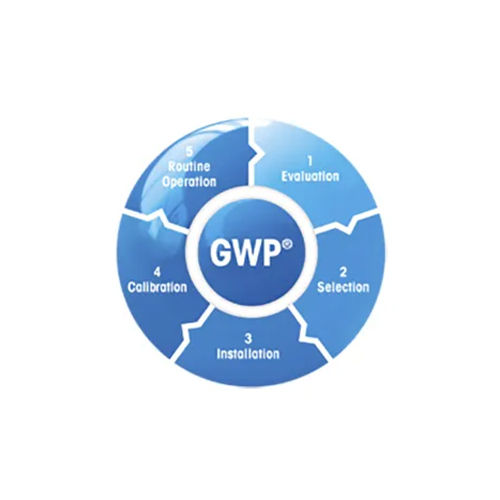
Calibration under GMP/USP/GWP:
GWP is a risk-based approach for managing weighing equipment, allows to improve control of the whole measuring process, which ensures reproducible product quality and minimizes process costs. GWP addresses the needs of all current quality management systems and puts them into weighing practice. It is the science-based standard for efficient life-cycle management of weighing equipment.
It fulfills all requirements of GMP to specify and calibrate balances and scales, independent of make or brand covering every relevant step in the equipment's life cycle.
It also addresses Metrology / Calibration, Quality Management, Process Requirements
Calibration with minimum weight certificate, Provides traceability- Determination of the measurement uncertainty of Instrument and minimum weight assures the instrument works within process requirements.
Certified Accuracy and Traceability
Each GMP/USP GC41,1058,1251 reference material is a certified reference standard, ensuring the highest levels of accuracy in laboratory calibration. Traceable to international standards, these materials provide consistent and reliable verification for equipment, supporting regulatory compliance and quality assurance programs.
Tailored Forms and Quantities
The product is available in both liquid and solid forms to match specific laboratory requirements. Customized packing options allow users to select the precise volume or quantity needed for their operational demands, minimizing waste and maximizing efficiency in laboratory management.
Optimal Storage and Extended Shelf Life
Strict storage recommendationsbetween 2C and 8C, protected from lightpreserve the integrity of the calibration and verification materials. With a shelf life of 24 months from the date of manufacture, laboratories can depend on lasting quality and consistent performance per batch.
FAQs of Calibration & Verification - GMPUSP GC41,1058, 1251:
Q: How should Calibration & Verification - GMP/USP GC41,1058,1251 materials be stored for best results?
A: These reference materials should be stored between 2C and 8C and kept away from light to maintain their integrity. Proper storage ensures the material remains effective throughout its 24-month shelf life.Q: What forms and packing options are available for this product?
A: The product is available in both liquid and solid forms, with customized packaging options to provide the required volume or quantity specific to your laboratory needs.Q: When is the recommended time to perform equipment calibration and verification with these standards?
A: Calibration and verification should be performed routinely, following your laboratorys SOPs or regulatory guidelines, and whenever new equipment is installed, after repairs, or when results appear inconsistent.Q: Where can these calibration and verification materials be used?
A: These certified reference standards are suitable for use in analytical laboratories, manufacturing facilities, research institutions, and quality assurance departments across various industries in India.Q: What benefits do certified reference standards offer over non-certified materials?
A: Certified reference standards provide traceability to international benchmarks, ensuring reliable and accurate calibration. This helps meet compliance requirements and enhances confidence in analytical results.Q: How are the materials packaged to ensure safety and traceability?
A: All materials are delivered in secure, tamper-proof containers, clearly labeled with batch information for traceability and compliance.Q: Can these materials be exported or supplied in bulk for large operations?
A: Yes, as a manufacturer, supplier, and exporter, we offer flexible supply options, including bulk quantities, to accommodate the needs of dealers, retailers, and laboratories nationwide and internationally.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
Other Products in 'Service and Support' category
 |
Mettler-Toledo India Private Limited
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese